 去看看
去看看


第一章 背景
第一节 子宫颈癌的流行病学状况
一、全球子宫颈癌的流行状况
据世界卫生组织/国际癌症研究署(World Health Organization/International Agency for Research on Cancer,WHO/IARC)2012年数据显示,子宫颈癌已成为女性第四大恶性肿瘤,全球每年新发子宫颈癌病例约52.8万,死亡26.6万,其中大约85%的子宫颈癌发生在发展中国家,占发展中国家女性肿瘤的12.0%,而发达国家子宫颈癌仅占女性肿瘤的3.0%。发展中国家的子宫颈癌年龄标化死亡率平均为8.3/10万,发达国家总体较低,为3.3/10万。
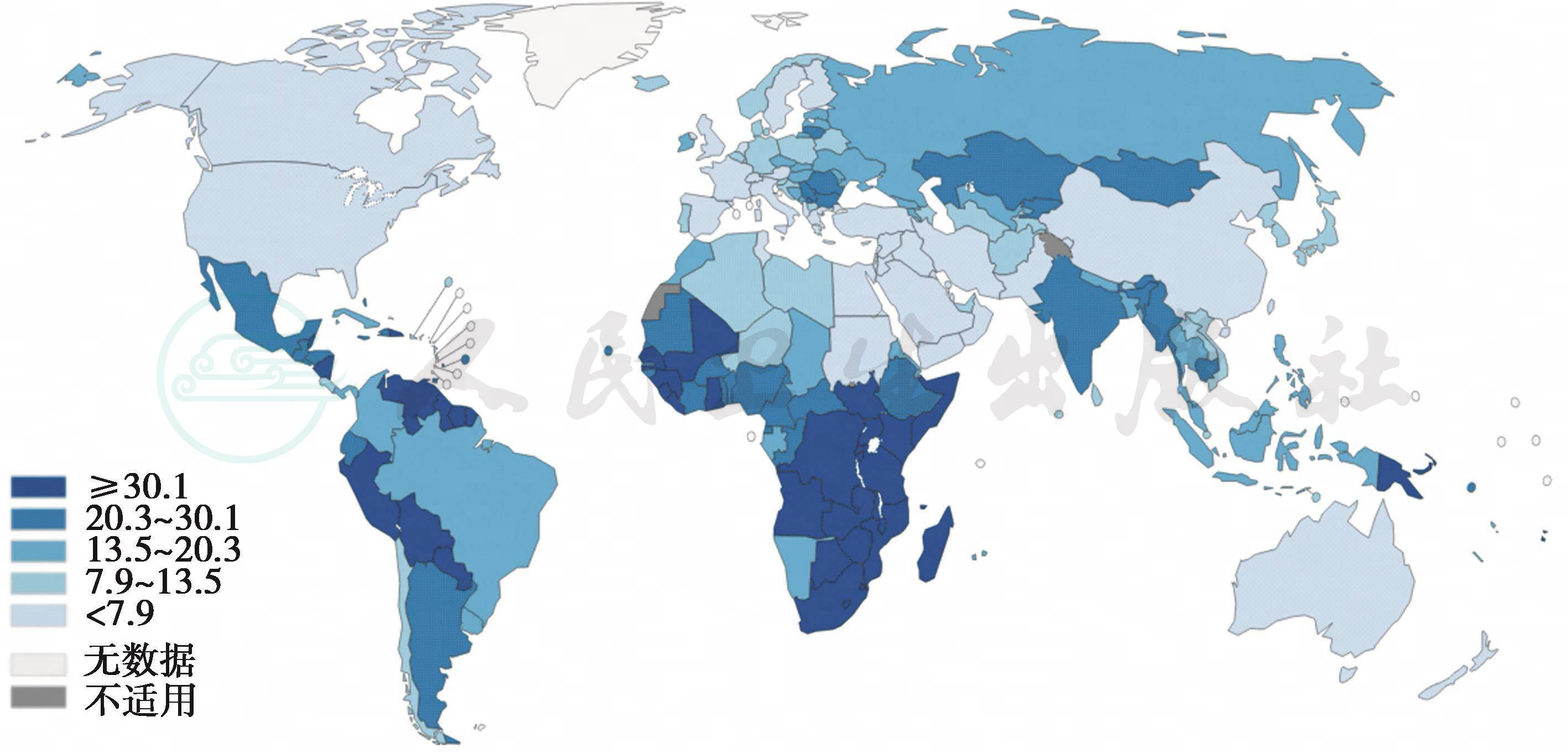
图1-1 子宫颈癌年龄标化发病??
第一章 背景
1.Ferlay J,Soerjomataram I,Ervik M,et al. FGLOBOCAN 2012 v1.0,Cancer Incidence and Mortality Worldwide:IARC CancerBase No. 11[Internet]. Lyon,France:International Agency for Research on Cancer;2013.Available from:http://globocan.iarc.fr,accessed on 15/5/2015.
2.Chen W,Zheng R,Baade PD,et al. Cancer statistics in China,2015.CA:a cancer journal for clinicians,2016,66(2):115-132.
3.胡尚英,郑荣寿,赵方辉,等. 1989至2008年中国女性子宫颈癌发病和死亡趋势分析.中国医学科学院学报,2014,36(2):119-125.
4.陈万青,郑荣寿,张思维,等. 2012 年中国恶性肿瘤发病和死亡分析. 中国肿瘤,2016,25(1):1-8.
5.卫生部肿瘤防治研究办公室.中国恶性肿瘤死亡调查研究. 北京:人民卫生出版社,1980:130.
6.包鹤龄,刘韫宁,王临虹,等. 2006-2012年我国宫颈癌死亡情况与变化趋势分析. 中华流行病学杂志,2017,38(1):58-64
7.Chen W,Zhang X,Molijn A,et al. Human papillomavirus type-distribution in cervical cancer in China:the importance of HPV 16 and 18.Cancer Causes Control,2009,20(9):1705-1713.
8.IARC. Human Papillomaviruses. Lyon,France,2007.
9.Zhao FH,Lewkowitz AK,Hu SY,et al. Prevalence of human papillomavirus and cervical intraepithelial neoplasia in China:a pooled analysis of 17 population-based studies. Int J Cancer,2012,131(12):2929-2938.
10.Östör AG. Natural history of cervical intraepithelial neoplasia:a critical review. International Journal of Gynecological Pathology,1993,12(2):186-192.
第二章 子宫颈癌综合防控项目与管理
1.WHO. Comprehensive cervical cancer control A guide to essential practice. Geneva:WHO press,2006.
2.IARC. IARC handbooks of cancer prevention (Vol. 10). Geneva:IARC Press,2005b.
3.Arbyn M,Anttila A,Jordan J,et al. European Guidelines for Quality Assurance in Cervical Cancer Screening. Second edition—summary document. Annals of oncology:official journal of the European Society for Medical Oncology/ESMO,2010,21(3),448-458.doi:10.1093/annonc/ mdp471.
4.ACCP. Planning and Implementing Cervical Cancer Prevention and Control Programs-A manual for managers:ACCP. 2004.
5.WHO. Comprehensive cervical cancer control-A guide to essential practice Second edition. Geneva:WHO Press,2014.
6.WHO. WHO guidance note:Comprehensive cervical cancer prevention and control:a healthier future for girls and women. Geneva:WHO Press,2013.
7.王临虹,魏丽惠. 妇女常见病筛查技术指南.北京:人民卫生出版社,2013.
8.魏丽惠,吴久玲. 子宫颈癌检查质量保障及质量控制指南.北京:人民卫生出版社,2015.
9.国家卫生与计划生育委员会妇幼健康服务司. 农村妇女“两癌”检查项目管理方案( 2015年版). 2015.
10.毕蕙,赵更力. 子宫颈癌综合防控技术培训教程.北京:人民卫生出版社,2015.
第三章 社会动员、健康教育和咨询
1.马骁. 健康教育学. 北京:人民卫生出版社,2004.
2.罗家有,张静. 妇幼健康教育学. 北京:人民卫生出版社,2014.
3.WHO. Comprehensive cervical cancer control A guide to essential practice. Geneva:WHO press,2006.
4.WHO. Comprehensive cervical cancer control-A guide to essential practice Second edition. Geneva:WHO Press,2014.
5.WHO. Preparing for the introduction of HPV vaccines:Policy and programme guidance for countries. 2006.
第四章 HPV疫苗接种
1.WHO. Human papillomavirus vaccines:WHO position paper,2017,92,241-268.
2.Harper DM,Franco EL,Wheeler CM,et al. Sustained efficacy up to 4.5 years of a bivalent L1 virus-like particle vaccine against human papillomavirus types 16 and 18:follow-up from a randomised control trial. Lancet,2006,367(9518):1247-1255.
3.Paavonen J,Jenkins D,Bosch FX,et al. Efficacy of a prophylactic adjuvanted bivalent L1 virus-like-particle vaccine against infection with human papillomavirus types 16 and 18 in young women:an interim analysis of a phase III double-blind,randomised controlled trial. The Lancet,2007,369(9580):2161-2170.
4.Naud PS,Roteli-Martins CM,De Carvalho NS,et al. Sustained efficacy,immunogenicity,and safety of the HPV-16/18 AS04-adjuvanted vaccine:Final analysis of a long-term follow-up study up to 9.4 years post-vaccination. Human vaccines & immunotherapeutics,2014,10(8):2147-2162.
5.Zhu FC,Chen W,Hu YM,et al. Efficacy,immunogenicity and safety of the HPV-16/18 AS04-adjuvanted vaccine in healthy Chinese women aged 18-25 years:Results from a randomized controlled trial. International Journal of Cancer,2014,135(11):2612-2622.
6.Zhao FH,Tiggelaar SM,et al. A multi-center survey of age of sexual debut and sexual behavior in Chinese women:suggestions for optimal age of human papillomavirus vaccination in China. Cancer Epidemiol,2012,36(4):384-390.
7.Zhu FC,Hu SY,Hong Y,et al. Efficacy,immunogenicity and safety of the HPV-16/18 AS04-adjuvanted vaccine in Chinese women aged 18-25 years:event-triggered analysis of a randomized controlled trial. Cancer Medicine,2017,6(1):12-25.
8.Schiller JT,Müller M. Next generation prophylactic human papillomavirus vaccines. The Lancet Oncology,2015,16(5):e217-e225.
9.WHO. Comprehensive cervical cancer control-A guide to essential practice Second edition. Geneva:WHO Press,2014.
10.国家卫生计生委《预防接种工作规范》. 2016版.
第五章 子宫颈癌筛查
1.Saslow D,Solomon D,Lawson HW,et al. American Cancer Society,American Society for Colposcopy and Cervical Pathology,and American Society for Clinical Pathology Screening Guidelines for the Prevention and Early Detection of Cervical Cancer.CA Cancer J Clin,2012,62(3):147-172.
2.乔友林,章文华,李凌,等. 子宫颈癌筛查方法的横断面比较研究. 中国医学科学院学报,2002,24(1):50-53.
3.Cuzick J,Clavel C,Petry KU,et al. Overview of the European and North American studies on HPV testing in primary cervical cancer screening. Int J Cancer,2006,119:1095-1101.
4.National Health Service. NHS cervical screening programmes. London:NHS,2014[2014-07-18].http://www.cancerscreening.nhs.uk/cervica/index.html.
5.Saslow D,Solomon D,Lawson HW,et al. American Cancer Society,American Society for Colposcopy and Cervical Pathology,and American Society for Clinical Pathology Screening Guidelines for the Prevention and Early Detection of Cervical Cancer.CA Cancer J Clin,2012,62(3):147-172.
6.Shi JF,Belinson JL,Zhao FH,et al. Human papillomavirus testing for cervical cancer screening:results from a 6-year prospective study in rural China. American journal of epidemiology,2009,170(6):708-716.
7.Zhao FH,Hu SY,Zhang Q,et al. Risk assessment to guide cervical screening strategies in a large Chinese population. Int J Cancer,2016,138(11):2639-2647.
8.Katki HA,Kinney WK,Fetterman B,et al. Cervical cancer risk for women undergoing concurrent testing for human papillomavirus and cervical cytology:a population-based study in routine clinical practice. Lancet Oncol,2011,12:663-672.
9.Sawaya GF,Smith-McCune K.Cervical Cancer Screening.OBSTETRICS & GYNECOLOGY,2016,127(3):459-462.
10.Hormuzd AK,Mark Schiffman,Philip EC,et al. Benchmarking CIN3+ risk as the basis for incorporating HPV and Pap cotesting into cervical screening and management guidelines. J Low Genit Tract Dis,2013,17:S28-S35.
11.Sawaya GF,Smith-McCune K.Cervical Cancer Screening.OBSTETRICS & GYNECOLOGY,2016,127(3):459-462.
第六章 子宫颈癌前病变的诊断及处理
1.L Stewart Massad,Mark H Einstein,Warner K Huh,et al. 2012 Updated Consensus Guidelines for the Management of Abnormal Cervical Cancer Screening Tests and Cancer precursors. Journal of Lower Genital Tract Disease,2013,17(5):S1-S27.
2. WHO. Comprehensive Cervical Cancer Control A guide to essential practice. 2nd edition.WHO,2014.
3.WHO. WHO guidelines for treatment of cervical intraepithelial neoplasia 2-3 and adenocarcinoma in situ:cryotherapy,large loop excision of the transformation zone,and cold knife conization.
4.WHO. WHO guidelines for screening and treatment of precancerous lesions for cervical cancer prevention(2013). Supplemental material:GRADE evidence-to-recommendation tables and evidence profiles for each recommendation.
5.WHO. Monitoring national cervical cancer prevention and control programmes:quality control and quality assurance for visual inspection with acetic acid (VIA)-based programmes (2013).
6.WHO. WHO guidelines:Use of cryotherapy for cervical intraepithelial neoplasia (2011).
7.Debbie Saslow,Diane Solomon,Herschel W Lawson,et al. American Cancer Society,American Society for Colposcopy and Cervical Pathology,and American Society for Clinical Pathology Screening Guidelines for the Prevention and Early Detection of Cervical Cancer. Cancer J Clin,2012,62:147-172.
8.Alan G Waxman,David Chelmow,Teresa M Darragh,et al. Revised Terminology for Cervical Histopathology and Its Implications for Management of High-Grade Squamous Intraepithelial Lesions of the Cervix Obstetrics & Gynecology,2012,120(6):1465-1471.
9.Bornstein J,Bentley J,Bosze P,et al. 2011 IFCPC colposcopic nomenclature. Obstet Gynecol,2012,120:166-172
10.中国优生科学协会阴道镜和宫颈病理学分会专家委员会.中国子宫颈癌筛查及异常管理相关问题专家共识(一).中国妇产科临床,2017,18(2):190-192.
11.中国优生科学协会阴道镜和宫颈病理学分会(CSCCP)专家委员会.中国子宫颈癌筛查及异常管理相关问题专家共识(二).中国妇产科临床,2017,18(3):286-288.
第七章 子宫颈浸润癌的诊断与处理
1.WHO.WHO guidelines for the pharmacological treatment of persisting pain in children with medical illness. Geneva:WHO,2012.
2.Ensuring balance in national policies on controlled substances:guidance for availability and accessibility of controlled medicines. Geneva:WHO,2011.
3.World Health Organization (WHO). Resolution WHA 67.19.Strengthening of palliative care as a component of integrated treatment throughout the life course. Sixty-seventh World Health Assembly,agenda item 15.5,Geneva,24 May 2014.Geneva:WHO,2014 (WHA)67.
4.马丁,沈铿,崔恒.常见妇科恶性肿瘤诊治指南.第5版. 北京:人民卫生出版社,2016.
5.Barakat RR,Berchuck A,Markman M,et al. Principles and practice of gynecologic oncology,6th edition. Philadelphia (PA):Wolters Kluwer/Lippincott Williams & Wilkins,2013.
6.Edge S,Byrd DR,Compton CC,et al. AJCC Cancer Staging Manual,7th edition. New York (NY):Springer,2010:395-402.
7.Gold MA,Tian C,Whitney CW,et al. Surgical versus radiographic determination of paraaortic lymph node metastases before chemoradiation for locally advanced cervical carcinoma:a Gynecologic Oncology Group Study. Cancer,2008,112(9):1954-1963.
8.Novetsky AP,Kuroki LM,Massad LS,et al. The utility and management of vaginal cytology after treatment for endometrial cancer. Obstet Gynecol,2013,121:129-135.
9.Pecorelli S. Revised FIGO staging for carcinoma of the vulva,cervix,and endometrium. Int J Gynaecol Obstet,2009,105(2):103-104.
10.Rimel BJ,Ferda A,Erwin J,et al. Cervicovaginal cytology in the detection of recurrence after cervical cancer treatment. Obstet Gynecol,2011,118(3):548-553.
11.Wiebe E,Denny L,Thomas G. Cancer of the cervix uteri. Int J Gynaecol Obstet,2012,119(Suppl 2):S100-109.doi:10.1016/S0020-7292(12)60023-X.